(C) 2012 G. Targino Valente. This is an open access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC-BY), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
Cichlids represent one of the most species-rich families of fishes and have attracted the attention of evolutionary biologists due to the rapid radiation occurring in some groups and the importance of some species in the world aquaculture. Cytogenetic analysis was conducted in 10 cichlid species from the Araguaia River, Amazon Basin, Brazil. The chromosome number was 2n=48 for all analyzed species except for Laetacara araguaiae Ottoni et Costa, 2009 (2n=44). Chromosomal polymorphism was detected only in Geophagus proximus (Castelnau, 1855), which exhibits an extra large submetacentric and and a dot-like chromosomes. Moreover, the C-banding revealed a general pericentromeric heterochromatic pattern and some additional blocks for some species. The heterochromatic blocks corresponding to AgNOR bearing regions were observed in all species and also corresponded to CMA3 positive blocks, which were observed in terminal regions. Besides the general conserved chromosomal and heterochromatin patterns for South American cichlids, the presence of GC-rich heterochromatin was quite different in the species Biotodoma cupido (Heckel, 1840), Geophagus proximus, Retroculus lapidifer (Castelnau, 1855), Crenicichla strigata Günther, 1862 and Heros efasciatus Heckel, 1840. The results suggest that independent events of heterochromatin modification occurred during chromosome evolution in the group, regardless of the conservation of macro-chromosomal structure.
chromosome evolution, fish chromosomes, genome, Cichlidae
The family Cichlidae includes more than 3000 species comprising one of the most species-rich families of vertebrates (
The African and Neotropical cichlids, Pseudocrenilabrinae and Cichlinae, respectively, are both monophyletic and represent sister groups (
The chromosome numbers of approximately 135 species of cichlids have been determined. Although more than 60% of the species present karyotypes with 2n=48, the diploid number ranges from 2n=32 to 2n=60 (
Synthesis of the cichlid species analyzed with respect to the karyotypic formulae, heterochromatin distribution and CMA3 patterns. m/sm, metacentric and submetacentric chromosomes; st/a, subtelocentric and acrocentric chromosomes; mi, microchromosomes; q, the long arm of a chromosome; p, the short arm of a chromosome; PeriC or C, pericentromeric regions; Prox, proximal portion of a chromosome; Term, Terminal portion of a chromosome; Int, interstitial portion of a chromosome; Adj, adjacent region; NOR, nucleolus organizing region; The numbers in the column “Additional blocks” indicate the number of chromosomes with the described pattern; in some cases, the ranking of these chromosomes are indicated in parentheses.
| Tribes and species | Origin of animals | 2n | Karyotypic formulae | Heterochromatin distribution | CMA3+ blocks | References | |
|---|---|---|---|---|---|---|---|
| General pattern | Additional blocks | ||||||
| Cichlini | |||||||
| Cichla piquiti Kullander et Ferreira, 2006 | Das Mortes river, Araguaia basin, MT State, Brazil | 48 | 48st/a | PeriC | NOR; term 2 | NOR (term) | This work |
| Cichla kelberi Kullander et Ferreira, 2006 | Araguaia river, MT State, Brazil | 48 | 48st/a | C | NOR; int 1 q | absent |
|
| Cichla monoculus Spix et Agassiz, 1831 | Uatumã and Solimões rivers, AM State, Brazil | 48 | 48a | PeriC | NOR; int 1 q | absent |
|
| Cichla temensis Humboldt, 1821 | Uatumã and Jaú rivers, AM State, Brazil | 48 | 48a | PeriC | NOR; int 1 q | absent |
|
| Retroculini | |||||||
| Retroculus lapidifer lapidifer (Castelnau, 1855) | Das Mortes river, Araguaia basin, MT State, Brazil | 48 | 48st/a | PeriC | NOR; term 1 q | NOR (term) and PeriC | This work |
| Astronotini | |||||||
| Astronotus ocellatus (Agassiz, 1831) | Tietê river, SP State, Brazil | 48 | 16m/sm + 32st/a | C | NOR | absent |
|
| Geophagini | |||||||
| Apistogramma trifasciata (Eigenmann et Kennedy, 1903) | Paraná river, Missiones, Argentina | 46 | 16m/sm + 30st/a | PeriC | absent | absent |
|
| Biotodoma cupido (Heckel, 1840) | Das Mortes river, Araguaia basin, MT State, Brazil | 48 | 4m/sm + 44st/a | PeriC | NOR; some prox blocks | NOR (int) | This work |
| Crenicichla britskii Kullander, 1982 | Jupiá river, PR State, Brazil | 48 | 8m/sm + 40st/a | PeriC | NOR; 1 p almost completely heterochromatic (1st pair) | absent |
|
| Crenicichla strigata Günther, 1862 | Das Mortes river, Araguaia basin, MT State, Brazil | 48 | 6m/sm + 42st/a | PeriC | NOR; some prox blocks | NOR (term) and PeriC | This work |
| Crenicichla prope johanna Heckel, 1840 | Negro and Solimões rivers, AM State, Brazil | 48 | 8m/sm + 40st/a | PeriC | NOR; term 1 q (19th pair) | absent |
|
| Crenicichla cincta Regan, 1905 | Negro and Solimões rivers, AM State, Brazil | 48 | 8m/sm + 40st/a | PeriC | adj NOR | absent |
|
| Crenicichla iguassuensis Haseman, 1911 | Iguaçu river, PR State, Brazil | 48 | 4m + 4sm + 14st + 26a | PeriC | Some term blocks | NOR |
|
| Crenicichla inpa Ploeg, 1991 | Negro and Solimões rives, AM State, Brazil | 48 | 6m/sm + 42st/a | PeriC | adj NOR | absent |
|
| Crenicichla lepidota Heckel, 1840 | São Gonçalo stream and Polegar lake, RS State, Brazil | 48 | 4m + 4sm + 40st/a | PeriC | term 1 p and 1 q (1st pair); int 1 q (1st pair) | NOR |
|
| Crenicichla lepidota Heckel, 1840 | Porto Rico region, Paraná river basin, PR State, Brazil | 48 | 2m + 4sm + 42st/a | PeriC | int 2 (1st and 5th pairs) | absent |
|
| Crenicichla lugubris Heckel, 1840 | Negro and Solimões rivers, AM State, Brazil | 48 | 8m/sm + 40st/a | PeriC | NOR; int 1 q (2nd pair) | absent |
|
| Crenicichla niederleinii (Holmberg, 1891) | Paraná river, Missiones, Argentina | 48 | 6m/sm + 42st/a | PeriC | absent | absent |
|
| Crenicichla reticulata (Heckel, 1840) | Negro and Solimões river, AM State, Brazil | 48 | 6m/sm + 42st/a | PeriC | adj NOR; int 1 q (10th pair) | absent |
|
| Crenicichla sp.1 | Iguaçu river, PR State, Brazil | 48 | 4m + 4sm + 14st + 26a | PeriC | Some term blocks | NOR |
|
| Crenicichla sp. 2 | Iguaçu river, PR State, Brazil | 48 | 4m + 4sm + 14st + 26a | PeriC | Some term blocks | NOR |
|
| Geophagus brasiliensis (Quoy et Gaimard, 1824) | Socavão and Verde rivers, PR State, Brazil | 48 | 6sm + 42st/a | PeriC/C | absent | NOR |
|
| Geophagus brasiliensis (Quoy et Gaimard, 1824) | Jaguarriaíva river, PR State, Brazil | 48 | 6sm + 42st/a | PeriC/C | Some int blocks | NOR |
|
| Geophagus brasiliensis (Quoy et Gaimard, 1824) | Saco da Alemoa, Gasômero, RS State, Brazil | 48 | 4sm + 44st/a | PeriC | NOR | NOR |
|
| Geophagus brasiliensis (Quoy et Gaimard, 1824) | Cambezinho and Três Bocas stream, Tibagi river basin, PR State, Brazil | 48 | 4sm + 44st/a | C | NOR | NOR |
|
| Geophagus brasiliensis (Quoy et Gaimard, 1824) | Pirapo river, Paranapanema basin, PR State, Brazil | 48 | 8sm + 40st/a | PeriC | prox 1 p (10th pair) | absent |
|
| Geophagus proximus (Castelnau, 1855) | Das Mortes river, Araguaia basin, MT State, Brazil | 48 | 4m/sm + 44st/a | PeriC | NOR; 1 p almost completely heterochromatic | NOR (int) | This work |
| Gymnogeophagus balzanii (Perugia, 1891) | Paraná river, Missiones State, Argentina | 48 | 2m/sm + 46st/a | PeriC | absent | absent |
|
| Geophagus gymnogenys (Hensel, 1870) | Saco da Alemoa, Barra do Ribeiro, Gasômetro, RS State, Brazil | 48 | 4m + 44st/a; 6m + 42st/a | PeriC | NOR | NOR |
|
| Geophagus labiatus (Hensel, 1870) | Saco da Alemoa, Forqueta river, RS State, Brazil | 48 | 4m + 4sm + 40st/a | PeriC | absent | NOR |
|
| Gymnogeophagus sp. | Paraná river, Missiones, Argentina | 48 | 2m/sm + 46st/a | PeriC | absent | absent |
|
| Satanoperca jurupari (Heckel, 1840) | Das Mortes river, Araguaia basin, MT State, Brazil | 48 | 4m/sm + 44st/a | PeriC | absent | NOR | This work |
| Satanoperca pappaterra (Heckel, 1840) | Porto rico region, Parana river basin, PR State, Brazil | 48 | 6sm + 42st/a | PeriC | absent | absent |
|
| Cichlasomatini | |||||||
| Aequidens tetramerus Heckel, 1840 | Araguaia river, MT State, Brazil | 48 | 12m/sm + 36st/a | PeriC | absent | NOR | This work |
| Australoheros facetus (Jenyns, 1842) | São Gonçalo stream and Polegar lake, RS State, Brazil | 48 | 22sm + 26st/a | PeriC/C | absent | NOR |
|
| Bujurquina vittata (Heckel, 1840) | Paraná river, Missiones, Argentina | 44 | 22m/sm + 8st/a + 14 mi | PeriC | NOR; p arm of 5th pair completely heterochromatic | absent |
|
| Cichlasoma dimerus (Heckel, 1840) | Paraná river, Missiones, Argentina | 48 | 8m/sm + 40st/a | PeriC | absent | absent |
|
| Cichlasoma facetum (Jenyns, 1842) | Tarumã lake, PR State, Brazil | 48 | 10sm + 38 st/a | PeriC/C | absent | NOR |
|
| Cichlasoma paranaense Kullander, 1983 | Porto rico region, Parana river basin, PR State, Brazil | 48 | 20sm + 28 st/a | PeriC | prox 2 p (2nd and 9th pairs) | absent |
|
| Laetacara araguaiae Ottoni et Costa, 2009 | Araguaia river, MT State, Brazil | 44 | 4m/sm + 40st/a | PeriC | absent | NOR | This work |
| Laetacara prope dorsigera (Heckel, 1840) | Paraná river, PR State, Brazil | 43 | 5m + 38a | C | NOR | absent |
|
| 44 | 4m + 40a | ||||||
| 45 | 3m + 42a | ||||||
| 46 | 2m + 44a | ||||||
| Heroini | |||||||
| Heros efasciatus Heckel, 1840 | Araguaia river, MT State, Brazil | 48 | 8m/sm + 40st/a | PeriC | absent | NOR (term) and int 1 p | This work |
| Mesonauta festivus (Heckel, 1840) | Das Mortes river, Araguaia basin, MT State, Brazil | 48 | 14m/sm + 34st/a | PeriC | NOR; term 2 q | NOR (term) | This work |
| Pterophyllum scalare (Schultze, 1823) | Jari river, PA State, Brazil | 48 | 12m/sm + 36st/a | PeriC/C | 1 p almost completely heterochromatic (1st pair) | NOR, some centromeres |
|
| Symphysodon aequifasciatus Pellegrin, 1904 | Bauana lake, Tefé river, AM State, Brazil | 60 | 8m/sm + 8st/a +4mi; 50m/sm + 6st/a +4mi | PeriC | Some prox blocks; int 1 q (1st pair) | absent |
|
| Symphysodon discus Heckel, 1840 | Boi-boi stream, Negro river, AM State, Brazil | 60 | 50m/sm + 10st/a; 54m/sm + 6st/a | PeriC | Some prox blocks | absent |
|
| Symphysodon haraldi Schultz, 1960 | Manacapuru river, AM State, Brazil | 60 | 52m/sm + 4st/a +4mi | PeriC | Some prox blocks | absent |
|
It was analyzed 10 South American cichlid species of the subfamily Cichlinae: Cichla piquiti Kullander et Ferreira, 2006 (4 individuals: sex not identified), Retroculus lapidifer (Castelnau, 1855) (6 individuals: 3 ♀ and 1 ♂, and 2 sex not identified), Biotodoma cupido (Heckel, 1840) (5 individuals: 2 ♀, and 3 ♂), Crenicichla strigata Günther, 1862 (12 individuals: 5 ♀, 5 ♂, and 2 sex not identified), Geophagus proximus (Castelnau, 1855) (9 individuals: 4 ♀, 2 ♂, and 3 sex not identified), Satanoperca jurupari (Heckel, 1840) (15 individuals: 7 ♀, 5 ♂, and 3 sex not identified), Aequidens tetramerus Heckel, 1840 (44 individuals: 21 ♀, 14 ♂, and 9 sex not identified), Laetacara araguaiae Ottoni et Costa, 2009 (5 individuals: 1 ♀, 1 ♂, and 3 sex not identified), Heros efasciatus Heckel, 1840 (5 individuals: 5 females) and Mesonauta festivus (Heckel, 1840) (5 individuals: 2 ♀, 1 ♂, and 2 sex not identified), which belong to the tribes Cichlini, Retroculini, Geophagini, Cichlasomatini and Heroini (Table 1). All individuals analyzed were not juveniles. Wild specimens were collected in several rivers that are part of the Araguaia River system, which is situated in the quadrant bounded by the coordinates 52°24'00"W, 15°30'S (DMS) and 52°05'00"W, 15°58'S (DMS) in the region of Barra do Garças, Mato Grosso State, Brazil. The sampling of wild animals was performed in accordance with Brazilian laws for environmental protection (wild collection permit, SISBIO/15729–1). The animals were maintained for 24 hours in an aired aquarium at a temperature ranging from 25°C to 28°C before collecting tissue samples. The fish were euthanized with a lethal dose of benzocaine followed by spinal section (Protocol 01204 – Committee of Ethical in Animal Experimentation – UNESP – São Paulo State University, Brazil) before removal of the kidneys for chromosome preparation.
Mitotic chromosome preparations were obtained from kidney cells according to
The chromosome structure was analyzed through silver nitrate staining, Chromomycin A3 (CMA3) staining and C-banding.
To detect nucleolus organizer regions (NORs), the silver staining of the chromosomes was performed according to
The constitutive heterochromatin was detected using saline solution according to
The CMA3 staining was conducted according to the method by
The chromosome spreads were analyzed using an Olympus BX 61 microscope, and the images were captured with the Olympus DP71 digital camera with the software Image-Pro MC 6.0. There were analyzed 30 metaphase spreads for all cytogenetic procedures performed for each animal sample. Karyotypes were arranged in the order of decreasing chromosome size, and the chromosomes were classified as either meta/submetacentrics (m/sm) or subtelo/acrocentrics (st/a).
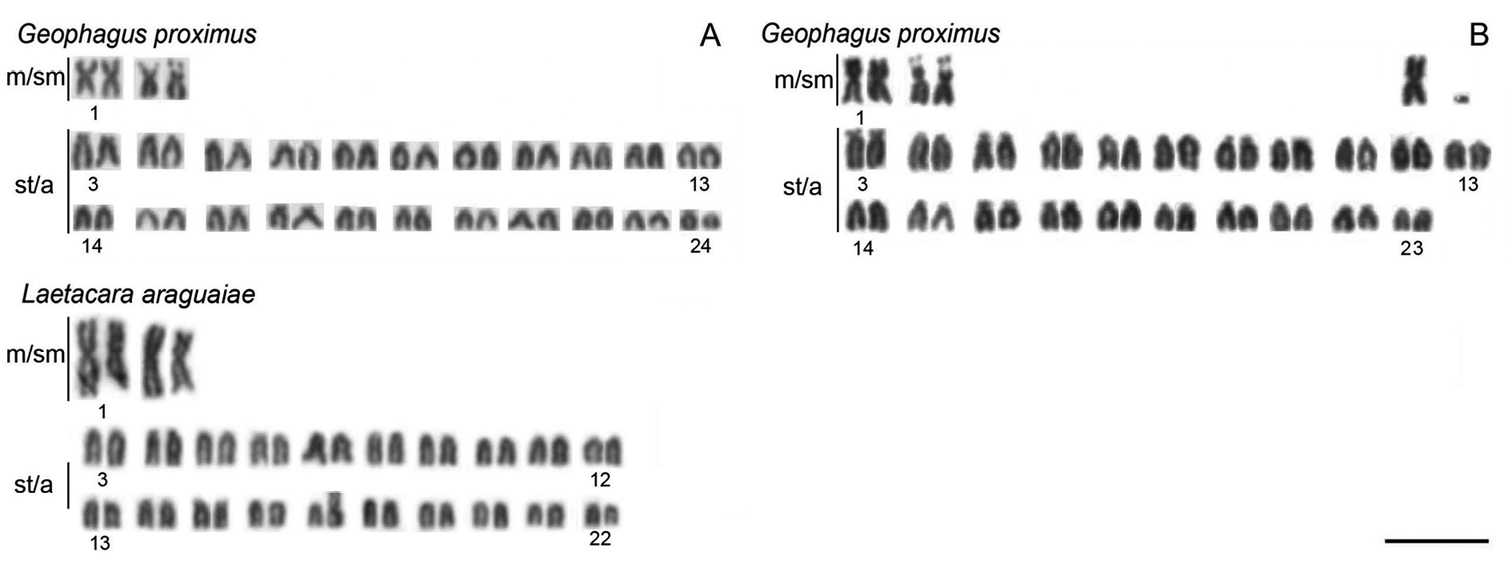
ResultsAll of the species analyzed have 2n=48 except Laetacara araguaiae, which showed a diploid number of 2n=44 and the karyotype formula of 4m/sm + 40st/a. Moreover, chromosomal polymorphism was found in Geophagus proximus, which presented two karyotype formulae, 4m/sm + 44st/a or 5m/sm + 42st/a + 1 dot-like chromosome (Fig. 1, Table 1).
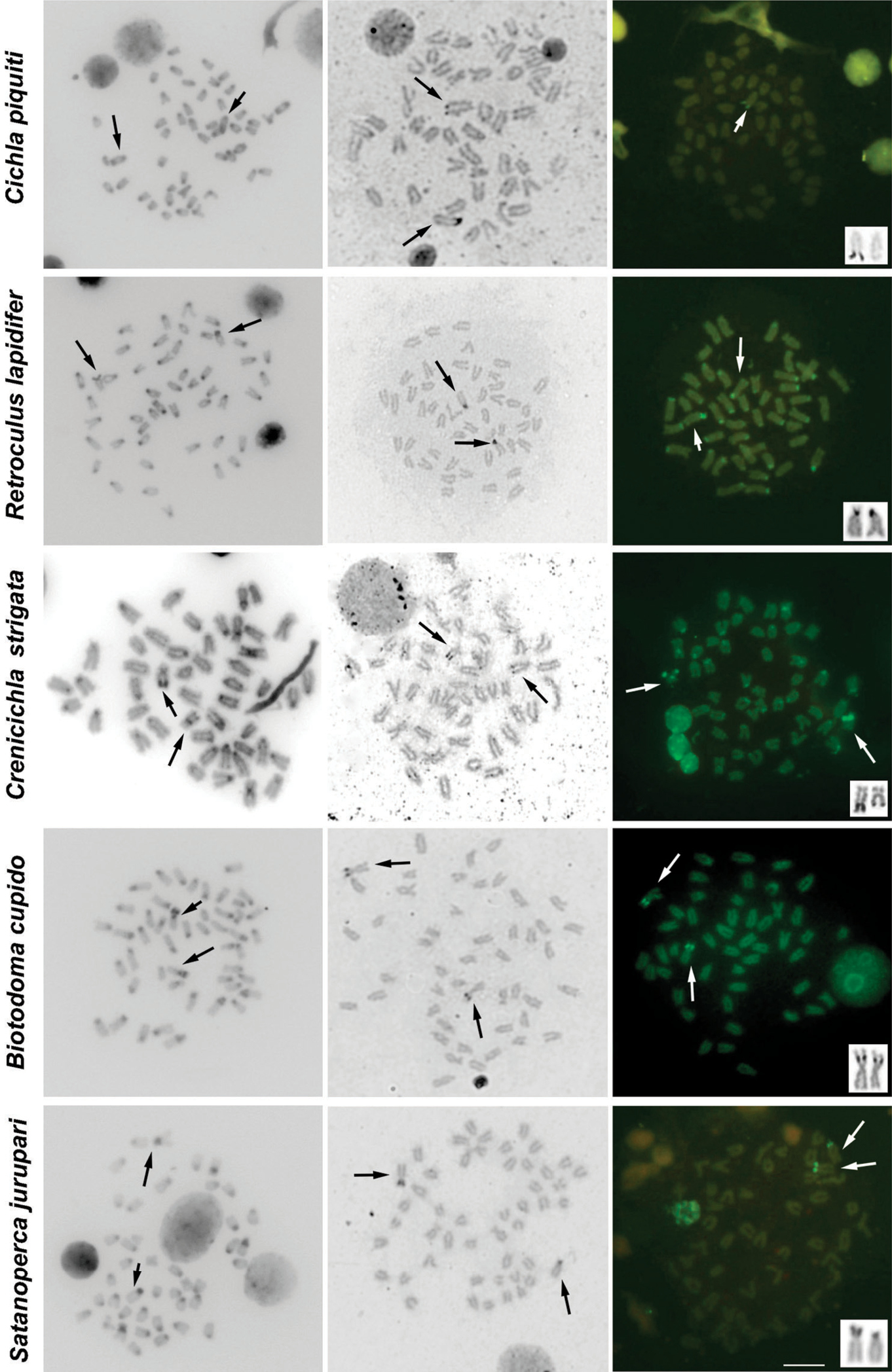
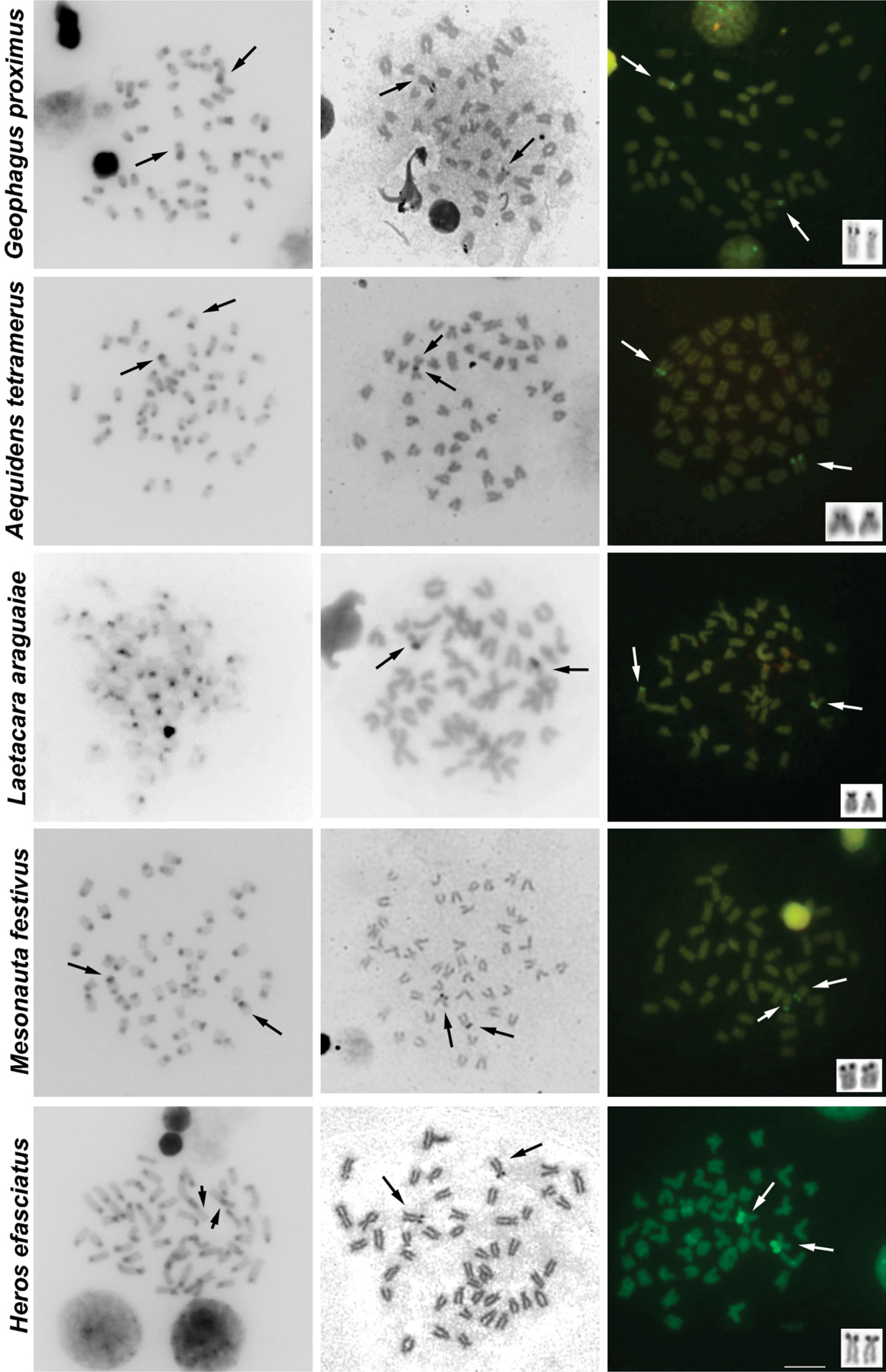
The results of C-banding revealed the heterochromatin generally restricted to pericentromeric regions. Additional blocks of heterochromatin were noticed in Cichla piquiti, Retroculus lapidifer, Biotodoma cupido, Crenicichla strigata, Geophagus proximus and Mesonauta festivus (Figs 2, 3, Table 1).
Characteristic heterochromatic blocks corresponding to AgNOR bearing regions (two blocks, one in each homologue) were observed in all species, and these blocks were consistent with CMA3 positive (CMA3+) blocks (Figs 2, 3, Table 1). These AgNOR/CMA3+ blocks were present in terminal regions; however, positional variation was observed in Biotodoma cupido (Fig. 2) and Geophagus proximus (Fig. 3), which the blocks are present in interstitial regions. Moreover, Retroculus lapidifer (Fig. 2) and Crenicichla strigata (Fig. 2) displayed CMA3+ blocks in pericentromeric regions of almost all chromosomes, and Heros efasciatus (Fig. 3) displayed a positive interstitial signal in one chromosome pair. Size variation was also observed in AgNOR/CMA3+ blocks between homologous chromosomes in Cichla piquiti (Fig. 2), Crenicichla strigata (Fig. 2) and Satanoperca jurupari (Fig. 2). Other chromosomal areas were CMA3 neutral in all of the species analyzed (Figs 2, 3).
Representative karyotypes of Geophagus proximus and Laetacara araguaiae species. For Geophagus proximus, two karyotypes are presented, a normal (A) and a polymorphic karyotype, showing in the upper right corner one extra large metacentric and one dot-like chromosome (B). Bar = 10 µm.
Metaphases of several cichlid species under different chromosome treatments. The species are indicated on the left. The first, second and third columns show C-banded, AgNOR- and CMA3- stained metaphases, respectively. The third column shows chromosomes bearing AgNORs in the box. The arrows indicate the NOR-bearing chromosomes. Bar = 10 μm.
Metaphases of several cichlid species under different chromosome treatments. The species are indicated on the left. The first, second and third columns show C-banded, AgNOR- and CMA3- stained metaphases, respectively. The third column shows chromosomes bearing AgNORs in the box. The arrows indicate the NOR-bearing chromosomes. For some metaphases (without arrows) it was not possible to identify the NOR-carrying chromosomes. Bar = 10 µm.
The diploid number reported for the species in this study, in general are in agreement with the conserved 2n=48 chromosomes commonly found in South American cichlids and in contrast with the presence of 2n=44 chromosomes in African cichlids. All species, except Laetacara araguaiae, had their diploid number already described (
Chromosomal variability was observed in derived lineages, such as the Geophagini and the Cichlasomatini tribes (
Chromosomal rearrangements such the ones reported here could lead to the karyotypic diversification of the species. In fact, chromosomal rearrangements have contributed to karyotypic evolution in a range of fishes, including the cichlids Symphysodon (Heckel, 1840) (
Although the cichlid cytogenetics suggests that the ancestral karyotype (2n=48 st/a) could have undergone major changes (pericentric inversions, fusions, fissions and chromosomal translocations) in the macro-structure of the South American species (
C-banding analyses in this study revealed that the conserved pattern of heterochromatin distribution was mostly restricted to the pericentromeric regions of cichlid chromosomes, which has been commonly reported in American and African representatives but with variations in both groups (
Concerning the singular heterochromatic blocks reported here, Cichla piquiti, Crenicichla strigata and Geophagus proximus show variability in the positions, extensions and number of these blocks compared to the other species in each genus. Moreover, the divergent patterns are observed in Crenicichla Heckel, 1840 and Geophagus Heckel, 1840. This variability can be also observed in the Laetacara genus; in this case, Laetacara araguaiae does not have any additional heterochromatic blocks, whereas Laetacara prope dorsigera has heterochromatic NORs as additional blocks (
The fluorochrome CMA3 showed the presence of GC-rich blocks coinciding with AgNOR sites in all species, which is a common trait in cichlids. The variation in the extension of these blocks also matches the size variation in the AgNOR sites in some species. Additional CMA3+ blocks are uncommon patterns in cichlids species, but they have been reported here for some species. In addition, this trait has only been previously reported in the Heroini species Pterophyllum scalare (Schultze, 1823) (
With respect to AgNOR, length variation between homologous chromosomes could be explained by the duplication or deletion of 45S rDNA repeat units. All AgNOR sites in the species analyzed here are heterochromatic as aforementioned. The length variation detected and extensively observed in other organisms may be caused by the presence of repetitive sequences, errors during the replication process, unequal crossing-over (
The heterochromatin, CMA3+ blocks and AgNOR regions are classic cases of enriched repetitive elements regions, such as satellite DNA, transposable elements, and rDNA. Among cichlids, it has been reported that the pericentromeric regions, which are commonly evidenced by C-banding, are repositories for a great amount of repetitive elements, such as transposable elements (
The study was supported by Fundação de Amparo à Pesquisa do Estado de Mato Grosso (FAPEMAT), Fundação de Amparo a Pesquisa do Estado de São Paulo (FAPESP), Coordenadoria de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).