(C) 2012 Edson Lourenço da Silva. This is an open access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC-BY), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
Thoracocharax stellatus (Characiformes, Gasteropelecidae) is a small Neotropical species of fish, widely distributed in several rivers of South America. Evidence for karyotype heteromorphysm in populations from different geographical regions has been reported for this species. In this way, populations of Thoracocharax stellatus from the Paraguay River basin were cytogenetically characterized and the results were compared with other studies performed in the same species but from different basins. The results showed a diploid number of 2n = 54 for Thoracocharax stellatus, with chromosomes arranged in 6 metacentric (m), 6 submetacentric (sm), 2 subtelocentric (st) and 40 acrocentric (a), for both sexes, with a simple Nucleolus Organiser Region (NOR) system reported by the techniques of silver nitrate impregnation and fluorescent in situ hybridisation (FISH) using 18S rDNA sequences as probe. The distribution of constitutive heterochromatin, observed by the C-band technique and Chromomycin A3 staining showed great similarity among the analyzed populations and consists mainly of discrete blocks in the pericentromeric and telomeric regions of most chromosomes. The presence of female heterogamety was also observed indicating a ZZ/ZW system with W chromosome almost totally heterochromatic. The results also show cytogenetic diversity of the group and are useful to understand the mechanisms of karyotype evolution of the family.
Thoracocharax stellatus, Ag-NOR, C-band, FISH, 18S rDNA
The family Gasteropelecidae (Characiformes) comprises a group of small Neotropical fishes that inhabit rivers of the main Central and South America basins, with exception of the southeast of Brazil and Chile (
The first taxonomic study regarding this family was conducted by
Despite the reduced number of species, this family presents many taxonomic problems related to the difficulty of finding species-specific characters. Carnegiella marthae (Myers, 1927) e.g. have osteological differences related to anal fin rays among specimens from Orinoco and Negro rivers, and the specimens from Peruvian Amazon and Madeira Rivers (
Thus, considering its large distribution, an accurate analysis of specimens from different localities can reveal the existence of putative new species (
As regards to the cytogenetic aspects, the family Gasteropelecidae is relatively non-conserved, with diploid numbers varying from 2n=48 to 2n=54. The species studied until now are recognized by the presence of many subtelocentric and acrocentric chromosomes and almost all presenting one chromosome pair carrier the NOR, that can be variable only in some populations (
The genus Thoracocharax is the most widely distributed among the gasteropelecids, and is characterized by species that have distinct chromosomal and morphological characteristics in different geographic regions. Thoracocharax stellatus (Kner, 1858), for example, presents a considerable morphological polymorphism among populations, mainly due to the geographic isolation (
The karyotypic diversity observed in Thoracocharax stellatus comprises different diploid numbers and polymorphism in NOR (Nucleolus Organiser Region) number and different chromosomes involved in the sexual differentiation (ZZ/ZW system) (
Due to the cytogenetic diversity already observed for different Thoracocharax stellatus populations, the objective of this study was to describe the karyotypic structure of three populations from the Paraguay basin not studied yet and identify chromosomal markers for this species by establishing chromosomal variation patterns among different populations.
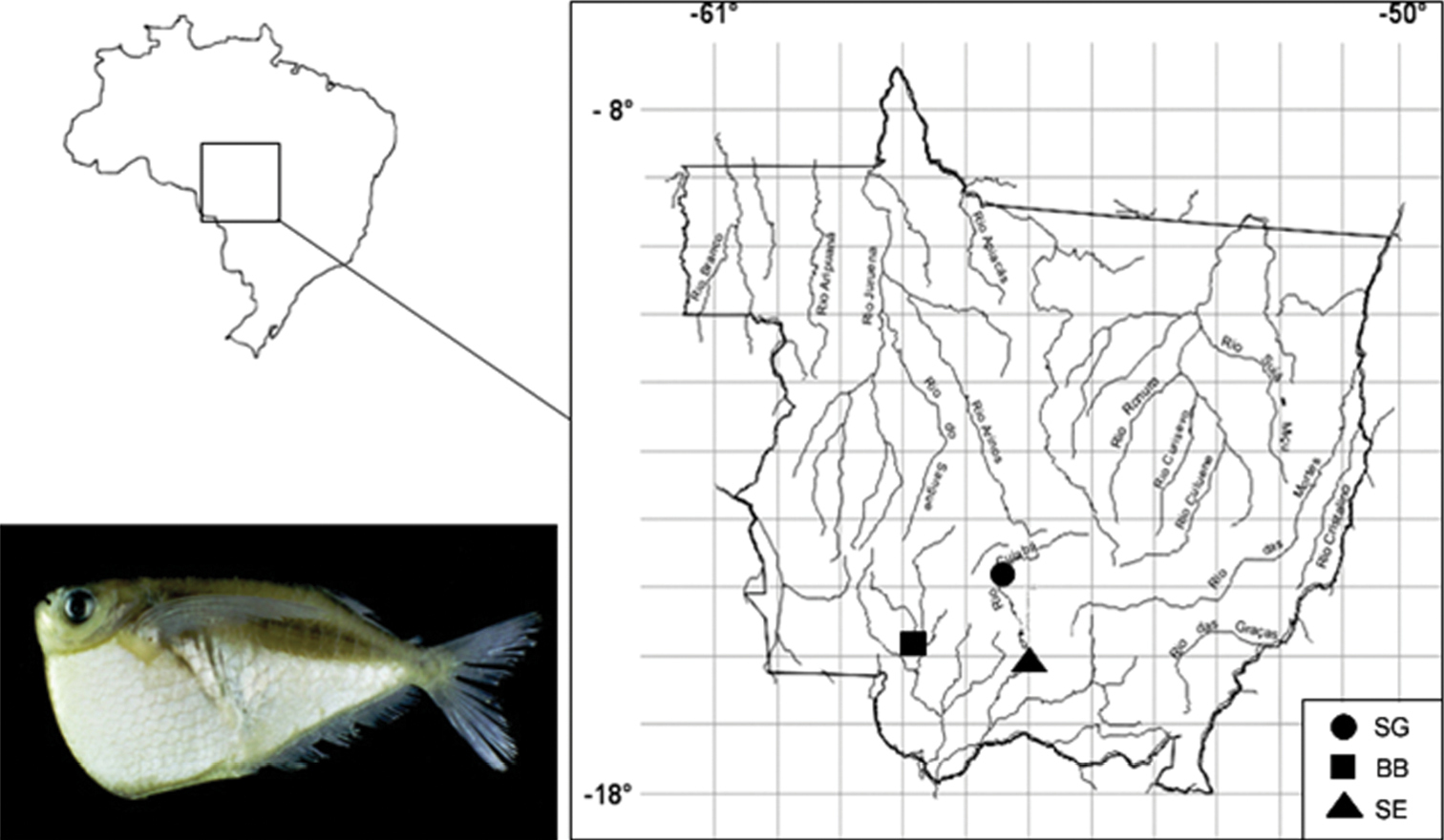
Material and methodsFor the cytogenetic analysis of Thoracocharax stellatus, 111 specimens collected in the Paraguay basin were used, 38 from Cuiabá River - São Gonçalo (SG) (15°39'9.96"S, 56°4'8.62"W), 26 in Cuiabá River - SESC (SE) (16°38'55.0"S, 56°28'06.2"W) and 47 from Barra do Bugres (BB) (15°4'41.13"S, 57°10'55.64"W) (Fig. 1). The material collected was deposited in the Laboratório de Citogenética Animal/UFMT, Mato Grosso (LCA 018, LCA 023, LCA 028).
Sampling sites of the three populations of Thoracocharax stellatus. SG: São Gonçalo; SE: SESC and BB Barra do Bugres.
Direct cytological preparations were produced from kidney samples according to the methods given by
The chromosomes were morphologically classified according to the nomenclature proposed by
The Fluorescent in situ hibridization (FISH) analyses were performed basically according to the method of
Chromosomes were observed using an Olympus BX51 microscope coupled to an Olympus digital camera model D71. Chromosome images were captured using the DP Controller software.
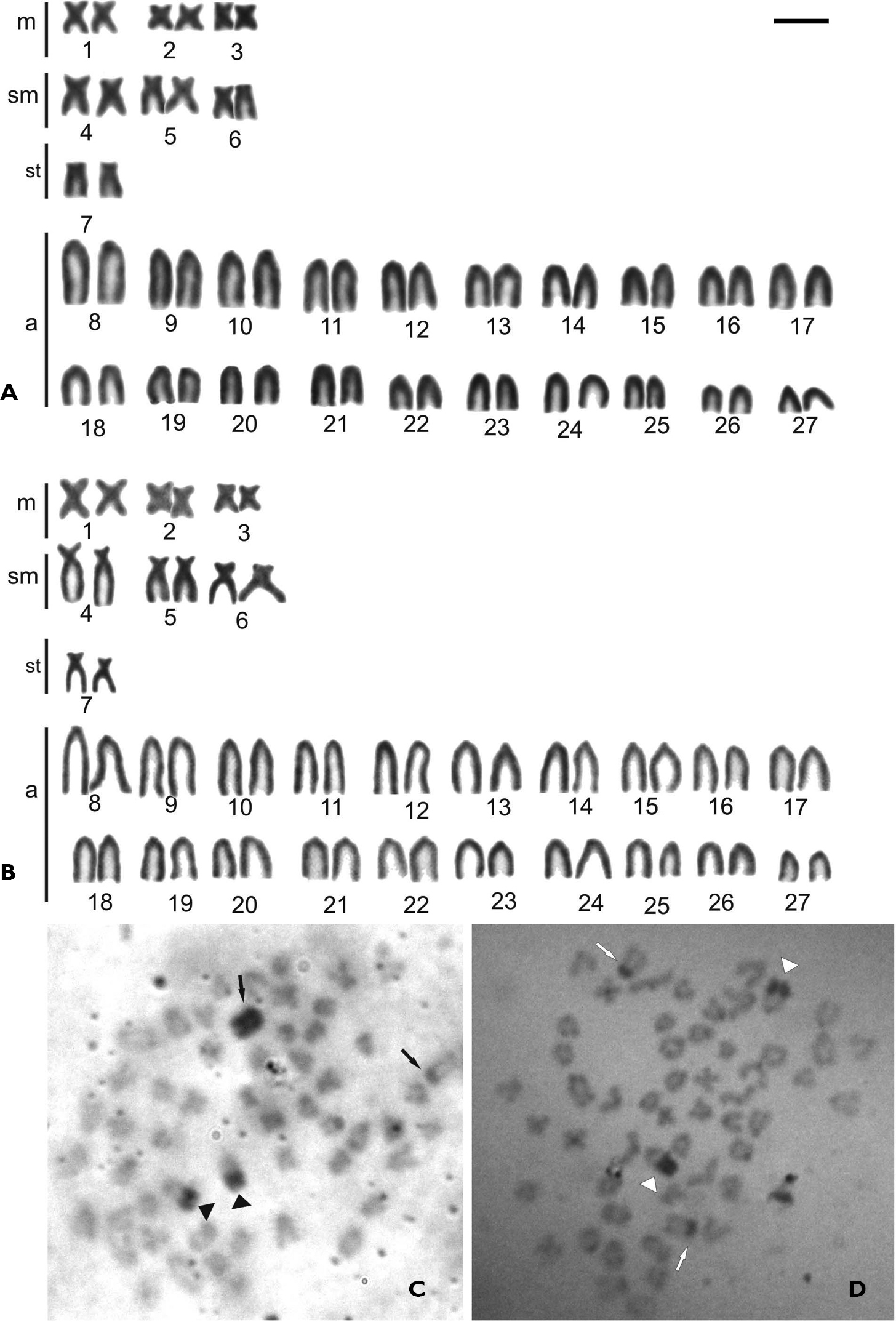
ResultsTable 1 summarizes the karyotypic data obtained in the present study as well as in the available literature. The Thoracocharax stellatus specimens from three localities of the Paraguay River basin present diploid number 2n=54 chromosomes, with fundamental number NF=68 and karyotypic formulae that includes 6m, 6sm, 2st and 40a for both sexes (Figure 2 A, B).
Chromosomal data on Gasteropelecidae from different Brazilian hydrographic basins.<br/>
| karyotypic formula | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Species | Hydrographical basin | Sex chromosomes | Ag-NOR | 2n | m | sm | st | a | Ref |
| Thoracocharax stellatus | Araguaia | ZZ/ZW | 2–4 | 54 | 6 | 6 | 6 | 36 | 1 |
| Thoracocharax stellatus | Paraguay | ZZ/ZW | 2 | 54 | 6 | 6 | 2 | 40 | 2 |
| Thoracocharax cf. stellatus | Amazon | ZZ/ZW | 2 ▲ | 52 | 8 | 16 | 4 | 24 | 3 |
| Carnegiella marthae | Amazon | ZZ/ZW | 2 | 50 | 20 | 12 | 4 | 14 | 4 |
| Carnegiella strigata | Amazon | - | 2–4 | 50 | 4 | 4 | 2 | 40 | 4 |
▲= Presence of size heteromorphisms. References= 1-
The distribution pattern of constitutive heterochromatin presents similarity for all populations and is composed by discrete blocks mainly at the telomeric and pericentromeric regions in the majority of the chromosomes (Figure 2 C, D). The only exception was one acrocentric chromosome totally heterochromatic present in female karyotype, while the other chromosome of the pair presents a remarkable marker only in the pericentromeric region.
The male individuals present the conspicuous heterochromatin blocks in the pericentromeric region in corresponding chromosomes, indicating the presence of ZZ/ZW chromosome sex system in the studied populations (Figure 2 C, D).
Female (A) and male (B) karyotype of Thoracocharax stellatus stained with Giemsa showing 2n=54 chromosomes. Metaphases from female (C) and male (D) of Thoracocharax stellatus showing heterochromatic blocks after C-band treatment. Arrows indicate the sex chromosomes Z and W in females and males Z and Z; arrowheads indicate the NOR chromosome. Bar = 10 µm.
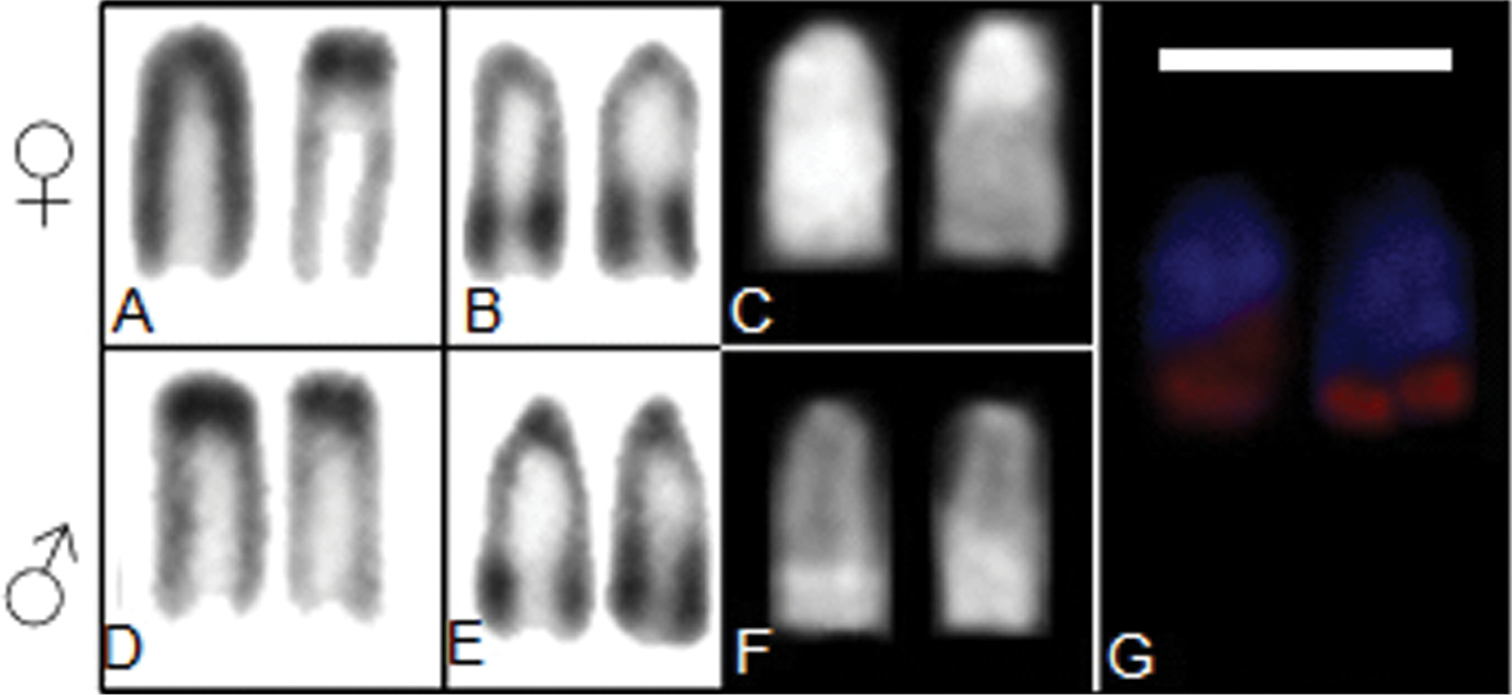
After colloidal silver nitrate treatment, was evidenced only one pair carrier the NOR. This structure is restricted to the terminal region of an acrocentric pair, confirmed by FISH technique (Figure 3 B, E, G). The heterochromatic segments of sex chromosomes, as well as NOR presents bright signals through Chromomycin A3 treatment, indicating a GC rich regions (Figure 3 C, F).
Chromosome markers from males and females individuals of Thoracocharax stellatus A C-band of Z and W chromosomes of female B Ag-NOR bearing chromosomes of female individuals C CMA3 treatment of Z and W chromosomes of female D C-band of ZZ chromosomes of male E Ag-NOR bearing chromosomes of male individuals F CMA3 treatment of ZZ chromosomes of male G FISH 18S rDNA. Bar = 10 µm.
In contrast to the situation observed in many families of the order Characiformes, the presence of non conserved karyotype is a common characteristic of Gasteropelecidae species. The diploid numbers range from 2n=48 to 2n=54 chromosomes (
The Thoracocharax stellatus populations analyzed in the present study presented 2n=54 chromosomes in both sexes, similar to the observed for individuals from Araguaia- Tocantins basin (
The NORs have been frequently employed as an important chromosome marker in many fish groups and their changes have been used as an important tool for the identification of chromosome markers among certain species and populations. The Thoracocharax stellatus populations studied presented a single NOR system, but a number and size heteromorphysm of these structures was detected in other populations already studied (Table 1). Considering that the karyotypic composition of the Paraguay and Araguaia basins is stable within each population, the variation in number of active sites in these populations can be explained by a possible differential expression of rDNA cistrons as observed in studies of other group of fishes in other basins (
According the results with base-specific fluorochromes the heterochromatin observed in Thoracocharax stellatus is distributed in blocks containing different proportions of the nucleotides. The first kind, present in few chromosomes of complement, is rich in GC bases. In this group, the heteromorphic chromosomes related to the ZZ/ZW sex system and the NOR chromosomes are included. The W chromosome has a large, CMA3+ positive heterochromatic fluorescent segment and in the Z only the pericentromeric regions is evidently heterochromatic. The sex chromosome system in Thoracocharax stellatus from the Paraguay basin does not present explicit differences in size between Z and W chromosomes, unlike that observed in populations from other hydrographic basins.
The second kind of heterochromatin, evidenced through C-band technique is characterized by markers located in pericentromeric and telomeric regions of several chromosomes of the complement, without fluorescent signals after the colouring with Chromomycin A3. This distribution pattern of heterochromatic blocks was already observed in several studies of chromosome characterization of many groups of fishes, as Leporinus (
The female heterogamety is the most frequently observed sex determination system observed in fishes (
Phylogenetic relationships based on morphological characters among gasteropelecids and other families have been discussed since
The increase of new cytogenetic studies have shown that other species of Gasteropelecidae such as Carnegiella marthae, have 50 chromosomes, NOR polymorphisms and a ZZ / ZW sex differentiation system (
Isolated events of chromosome rearrangements, without phylogenetic implications, as fission may be responsible to reduction of subtelocentric and increase of larges acrocentric chromosomes, resulting in the chromosomal particularities observed in Thoracocharax stellatus from Paraguay Rivers basin in relation to other population already studied. The results also show the cytogenetic diversity of the group, is useful to understand the mechanisms of karyotype evolution of the family as well as understanding the processes of differentiation and evolution of sex chromosomes in the group.
The authors would like to thank CAPES, FAPEMAT, BASA and UFMT, for the financial support.