(C) 2012 Eduard Petitpierre. This is an open access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC-BY), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
Four species of Chilean leaf beetles in the subfamily Chrysomelinae have been cytogenetically analyzed, Blaptea elguetai Petitpierre, 2011, Henicotherus porteri Bréthes, 1929 and Jolivetia obscura (Philippi, 1864) show 2n = 28 chromosomes and a 13 + Xyp male meioformula, and Pataya nitida (Philippi, 1864) has the highest number of 2n = 38 chromosomes. The karyotype of Henicotherus porteri is made of mostly small meta/submetacentric chromosomes, and that of Jolivetia obscura displays striking procentric blocks of heterochromatin at pachytene autosomic bivalents using conventional staining. These findings are discussed in relation to previous cytogenetic data and current taxonomy of the subfamily.
Coleoptera, Chrysomelidae, Chrysomelinae, karyotypes, cytotaxonomy
The subfamily Chrysomelinae is a group of mostly quite large or medium sized leaf beetles mainly distributed in cool and temperate regions of the world, which are composed of 133 genera (
From the cytogenetic standpoints, this subfamily is relatively well-known since nearly 260 taxa and chromosomal races in 38 genera have been surveyed to date (
The checked species and their origins are reported in Table 1.
Chromosomally checked species and their Chilean geographical sources.
| Blaptea elguetai Petitpierre, 2011 | Isla Negra, prov. San Antonio, Reg. Valparaíso |
| Henicotherus porteri Bréthes, 1929 | Mincha 2 km W, prov. Choapa, Reg. Coquimbo |
| Jolivetia obscura (Philippi, 1864) | Isla Negra, prov. San Antonio, Reg. Valparaíso |
| Pataya nitida (Philippi, 1864) | Isla Negra, prov. San Antonio, Reg. Valparaíso |
The surveyed individuals of Blaptea elguetai, Jolivetia obscura and Pataya nitida were caught by sweeping on their host plants Tropaeolum brachyceras Hook. et Arn., 1830 (Tropaeolaceae), Aristeguietia salvia (Colla) R.M. King et H. Rob., 1975 (Asteraceae) and Ageratina glechonophylla (Less.) R.M. King et H. Rob., 1970 (Asteraceae), in October 2009, October 2006 and November 2007, respectively, and those of Henicotherus porteri were caught by hand under stones in October 2009. At least two individuals from each species have been cytogenetically studied.
The chromosome analyses were only performed on male living individuals brought from Chile to our laboratory in Palma de Mallorca (Spain), where they were killed with ethyl acetate. Then, the procedure to get the conventional staining preparations was the same used before in our previous paper (
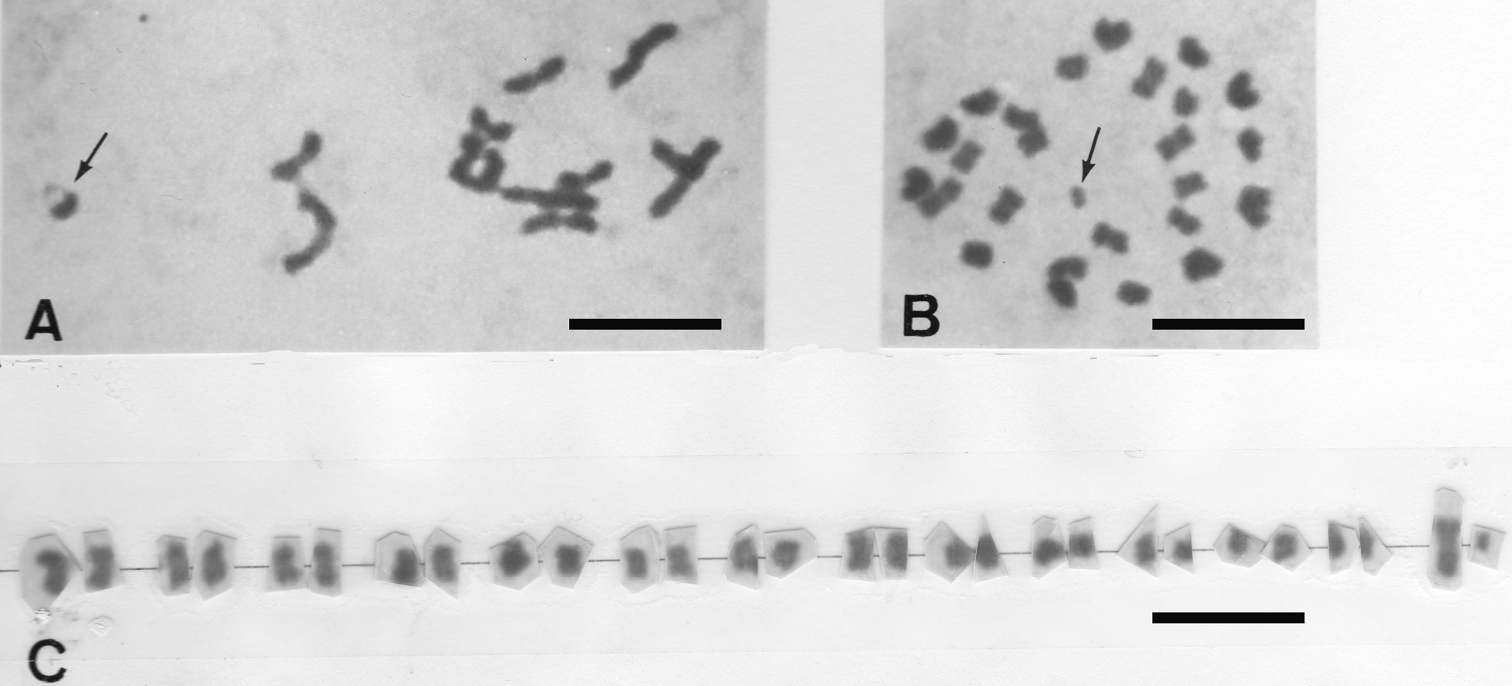
Blaptea elguetai has 2n = 28 chromosomes and a 13 + Xyp male meioformula of medium and small autosomal bivalents plus the Xyp “parachute” sex-chromosome system where most of these autosomic bivalents are rod-shaped (Fig. 1A).
Henicotherus porteri has also 2n = 28 chomosomes at spermatogonial metaphases (Fig. 1B), from which a karyogram has been obtained, made of medium and small metacentrics of gradually decreasing sizes, including the largest X-chromosome and the smallest y-chromosome elements (Fig. 1C). Confirming what was expected, the metaphases I comprise 13 autosomic bivalents and the Xyp sex-chromosome system (not shown).
A Blaptea elguetai metaphase I showing 13 + Xyp, the Xyp is arrowed B Henicotherus porteri spermatogonial metaphase with 2n = 28, the y-chromosome is arrowed.C karyogram showing small meta/submetacentric chromosome pairs, the medium-sized X and the smallest y-chromosome are in the extreme right. Bar = 10 µm
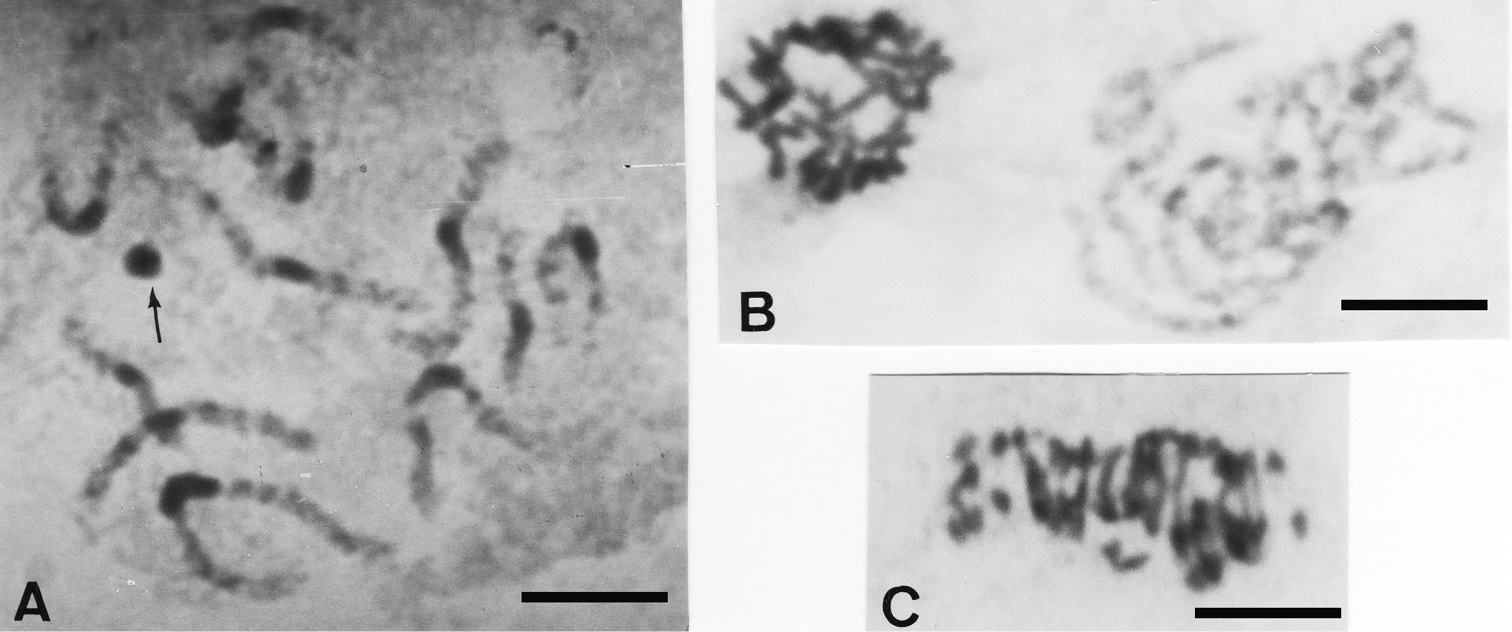
Jolivetia obscura displays 2n = 28 chromosomes as in the two previous species, from pachytene meiotic cells where 14 bivalents are distinguishable and among them the Xyp sex-chromosome system. Each of the 13 pachytene autosomal bivalents show a remarkable band of procentric heterocromatin, and the presumed Xyp sex-chromosome system appears as a strongly heterochromatic round bulk under the conventional staining technique (Fig. 2A).
Conversely, Pataya nitida displays a higher diploid number of 19 pachytene bivalents, a few of which having heterochromatic bands (Fig. 2B), and 2n = 38 small chromosomes at spermatogonial anaphase (Fig. 2C).
A Jolivetia obscura pachytene showing 13 + Xyp with striking procentric heterochromatic bands in the autosome bivalents, and the presumed Xyp arrowed B Pataya nitida spermatogonial metaphase (left) and pachytene (right), some autosome bivalents show small procentric bands of heterochromatin C anaphase I showing 2n = 38 chromosomes. Bar = 10 µm
The diploid number of chromosomes and male sex-chromosome system of Blaptea elguetai 2n = 28 (Xyp) agrees with our findings in Microtheca ochroloma Stål, 1860 (
Among the different subtribes of Chrysomelinae (
As reported above, Jolivetia obscura and Pataya nitida are classified in a different subtribe, Chrysomelina, than the two previous species (
The procentric bands of heterochromatin found in pachytene autosomal bivalents of Jolivetia obscura and in some of those of Pataya nitida, using conventional staining, are common feature in beetle chromosomes, as it has been recently demonstrated in several families of Coleoptera including Chrysomelidae by C-banding techniques (
The sex-chromosome system found in our sampled species of Chilean chrysomelines was the parachute-type Xyp, except in Pataya nitida which has not been identified. Thus, they agree with those found in the three previously analysed species of Chilean chrysomelines (
We are very much indebted to Marcelo Guerrero (Santiago, Chile) and Mauro Daccordi (Verona, Italy) who helped to the authors in collecting some of these four species in Isla Negra and Mincha, respectively. Miguel Angel Conesa (UIB, Palma de Mallorca) has given a valuable aid in arranging the micrographs.