Citation: Yano CF, Bertollo LAC, Molina WF, Liehr T, Cioffi MB (2014) Genomic organization of repetitive DNAs and its implications for male karyotype and the neo-Y chromosome differentiation in Erythrinus erythrinus (Characiformes, Erythrinidae). Comparative Cytogenetics 8(2): 139–151. doi: 10.3897/CompCytogen.v8i2.7597
Studies have demonstrated the effective participation of repetitive DNA sequences in the origin and differentiation of the sex chromosomes in some biological groups. In this study several microsatellites and retrotranposable sequences were cytogenetically mapped in the Erythrinus erythrinus (Bloch & Schneider, 1801) male genome (karyomorph C), focusing on the distribution of these sequences in the sex chromosomes and in the evolutionary processes related to their differentiation. Males of E. erythrinus – karyomorph C – present 2n = 51 chromosomes (7m + 2sm + 6st + 36a), including the X1X2Y sex chromosomes. The C-positive heterochromatin has a predominant localization on the centromeric region of most chromosome pairs, but also in some telomeric regions. The 5S rDNA sites are located in the centromeric region of 27 chromosomes, including 26 acrocentric ones and the metacentric Y chromosome. The retrotransposons Rex 1 and Rex 6 show a dispersed pattern in the karyotype, contrasting with the Rex 3 distribution which is clearly co-localized with all the 27 5S rDNA sites. The microsatellite sequences show a differential distribution, some of them restricted to telomeric and/or interstitial regions and others with a scattered distribution on the chromosomes. However, no preferential accumulation of these elements were observed in the neo-Y chromosome, in contrast to what usually occurs in simple sex chromosome systems.
FISH, microsatellites, retrotransposable sequences, sex chromosomes
Sex chromosomes have been widely studied in several invertebrate, vertebrate and plant individuals, focusing on their origin and differentiation (
Repetitive sequences include different classes of in tandem repeats, such as satellite DNAs, minisatellites and microsatellites, and interspersed repeats, like the transposable elements (TEs) (
Erythrinidae (Characiformes), are a small Neotropical fish family composed of three genera, Erythrinus Scopoli, 1777, Hoplerythrinus Gill, 1896 and Hoplias Gill, 1903 (
In this study several microsatellites and retrotransposable sequences were cytogenetically mapped in the Erythrinus erythrinus male genome (karyomorph C), focusing on their distribution in the sex chromosomes and in the evolutionary processes related to the differentiation of the neo-Y chromosome.
Six male specimens of Erythrinus erythrinus (karyomorph C), from the Manaus region (3°13'41.4"S, 59°43'43.1"W – Amazon State, Brazil) were analyzed. The experiments followed ethical conducts, and anesthesia was used prior to sacrificing the animals. Mitotic chromosomes were obtained from the anterior portion of the kidney, according to
Oligonucleotide probes containing microsatellite sequences (CA)15, (CAA)10, (CAC)10, (CAG)10, (CAT)10, (CGG)10, (GA)15, (GAA)10, (GAG)10 and (TA)15 were directly labeled with Cy5 during synthesis by Sigma (St. Louis, MO, USA), as described by
The FISH method was conducted as follows: slides with fixed chromosomes were maintained at 37 °C for 1 hour. Subsequently, they were incubated with RNAse (10 mg/ml) for 1 hour at 37 °C in a moist chamber. Next, it was performed a 5-minute wash with 1xPBS and 0.005% pepsin was applied to the slides (10 minutes at room temperature). The slides were then washed again with 1xPBS. The material was fixed with 1% formaldehyde at room temperature for 10 minutes. After further washing, the slides were dehydrated with 70%, 85% and 100% ethanol, 2 minutes in each bath. The chromosomal DNA was denatured in 70% formamide/2xSSC for 3 minutes at 72 °C. The slides were dehydrated again in a cold ethanol series (70%, 85% and 100%), 5 min each. The hybridization mixture, containing 100 ng of denatured probe, 10 mg/ml dextran sulfate, 2xSSC and 50% formamide (final volume of 30 μl) were heated to 95 °C for 10 minutes and then applied on the slides. Hybridization was performed for a period of 16-18 hours at 37 °C in a moist chamber. After hybridization, the slides were washed for 5 minutes with 2xSSC and then rinsed quickly in 1xPBS. The signal detection was performed using anti-digoxigenin rhodamine (Roche) for the 5S rDNA, Rex 1, Rex 3 and Rex 6 probes. Subsequently, the slides were dehydrated again in an ethanol series (70%, 85% and 100%), 2 minutes each. After the complete drying of the slides, the chromosomes were counterstained with DAPI/antifade (1.2 mg/ml, Vector Laboratories).
Approximately 30 metaphase spreads were analyzed to confirm the diploid chromosome number, karyotype structure and FISH results. Images were captured by the CoolSNAP system software, Image Pro Plus, 4.1 (Media Cybernetics, Silver Spring, MD, USA), coupled to an Olympus BX50 microscope (Olympus Corporation, Ishikawa, Japan). The chromosomes were classified as metacentric (m), submetacentric (sm), subtelocentric (st) or acrocentic (a), according to their arm ratios (
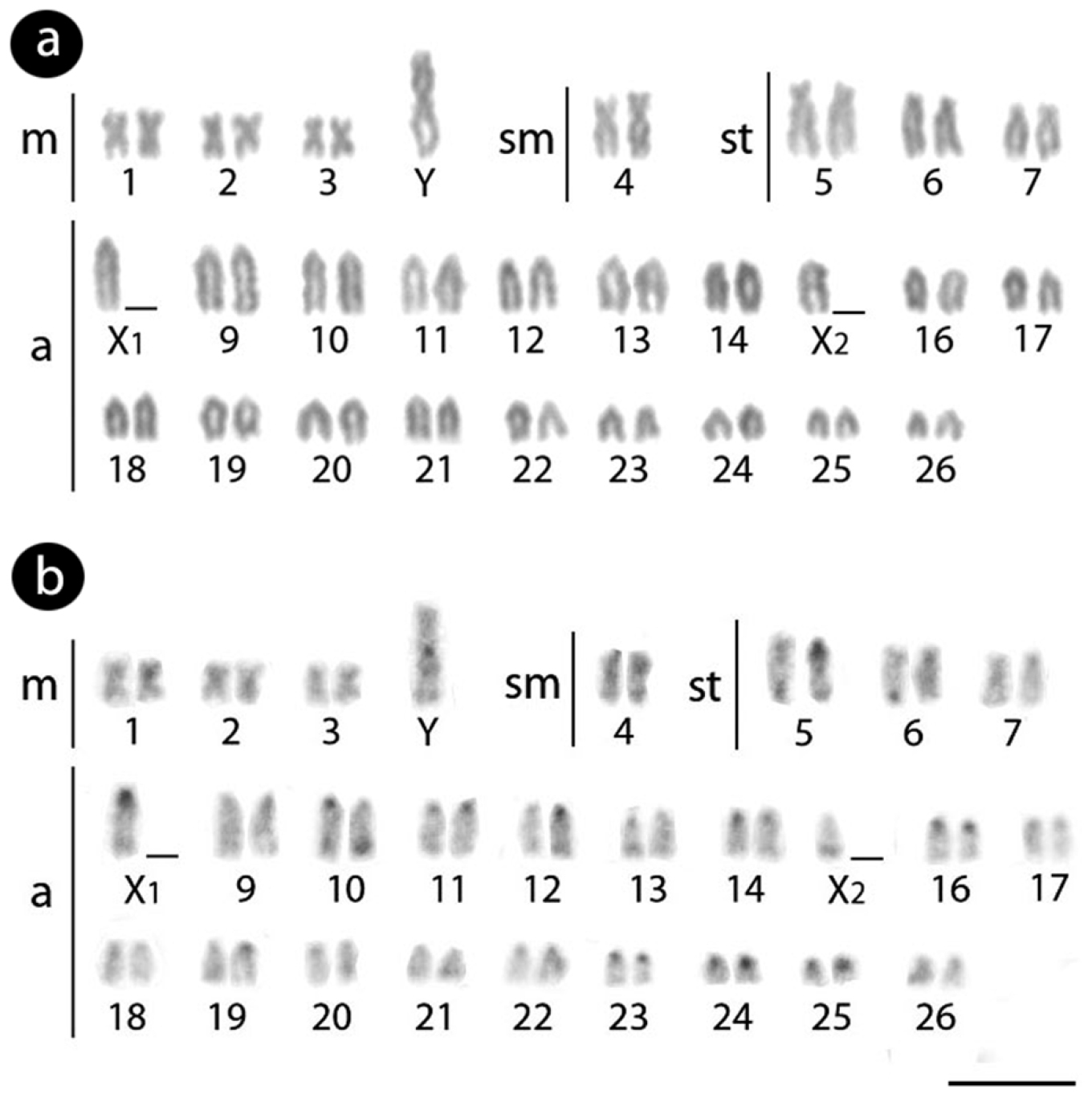
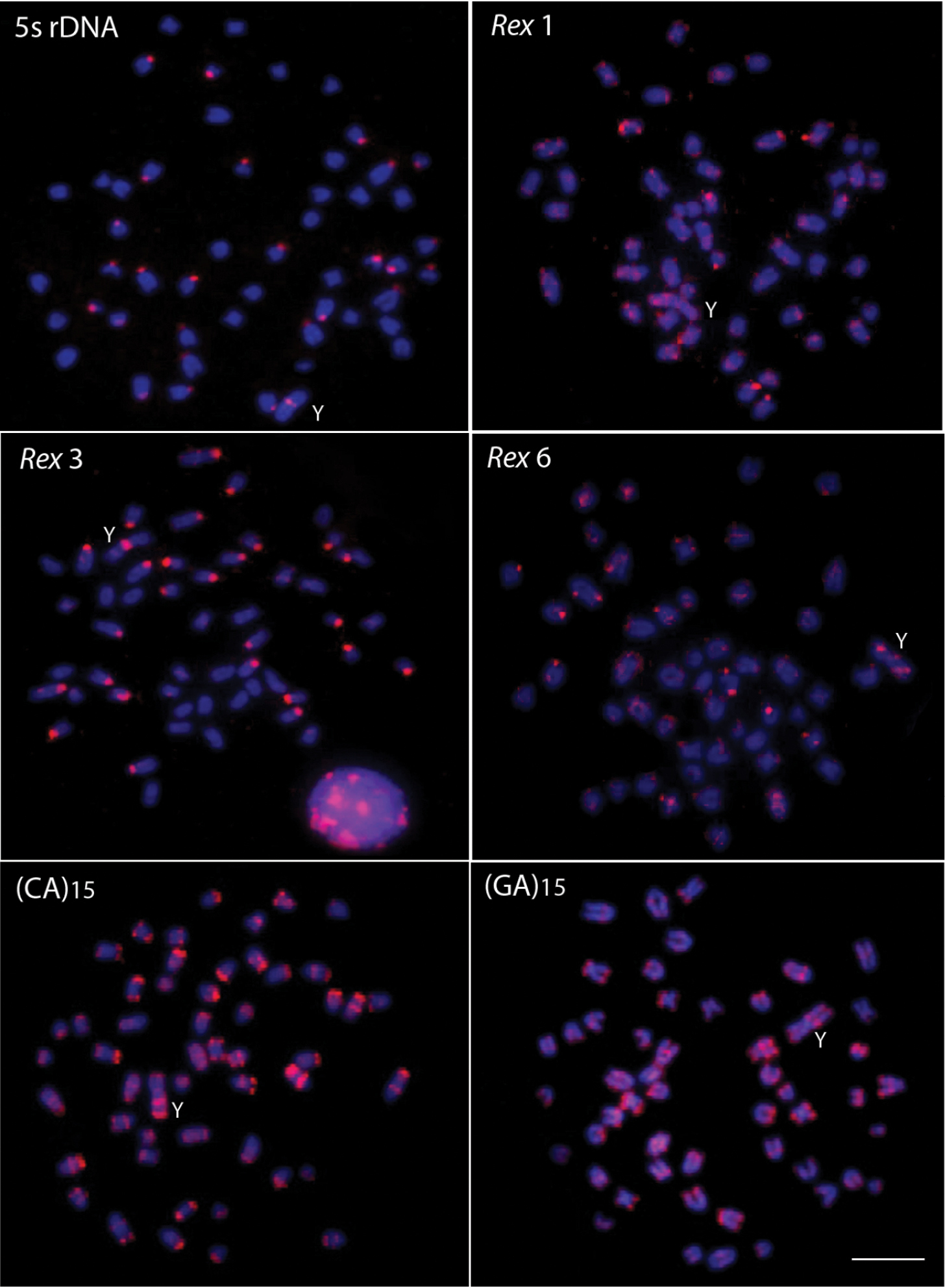
Males of Erythrinus erythrinus – karyomorph C – present 2n = 51 chromosomes (7m + 2sm + 6st + 36a), including the X1X2Y sex chromosomes. While the chromosomes X1 and X2 are acrocentric, the Y is the largest metacentric chromosome in the karyotype (Fig. 1a). The C-positive heterochromatin has a predominant localization on the centromeric region of most chromosome pairs, but also in some telomeric regions (Fig. 1b). The 5S rDNA sites are located in the centromeric region of 27 chromosomes, including 26 acrocentric ones and the metacentric Y-chromosome (Fig. 2). The retrotransposons Rex 1 and Rex 6 show a dispersed pattern in the karyotype, contrasting with the Rex 3 distribution which is clearly co-localized with all the 27 5S rDNA sites (Fig. 2).
Male Karyotype of Erythrinus erythrinus arranged from Giemsa-stained (a) and C-banded chromosomes (b). Bar = 5 µm.
Male metaphase plates of Erythrinus erythrinus probed with 5S rDNA, Rex 1, Rex 3 and Rex 6 transposons and microsatellite sequences. Bar = 5 μm.
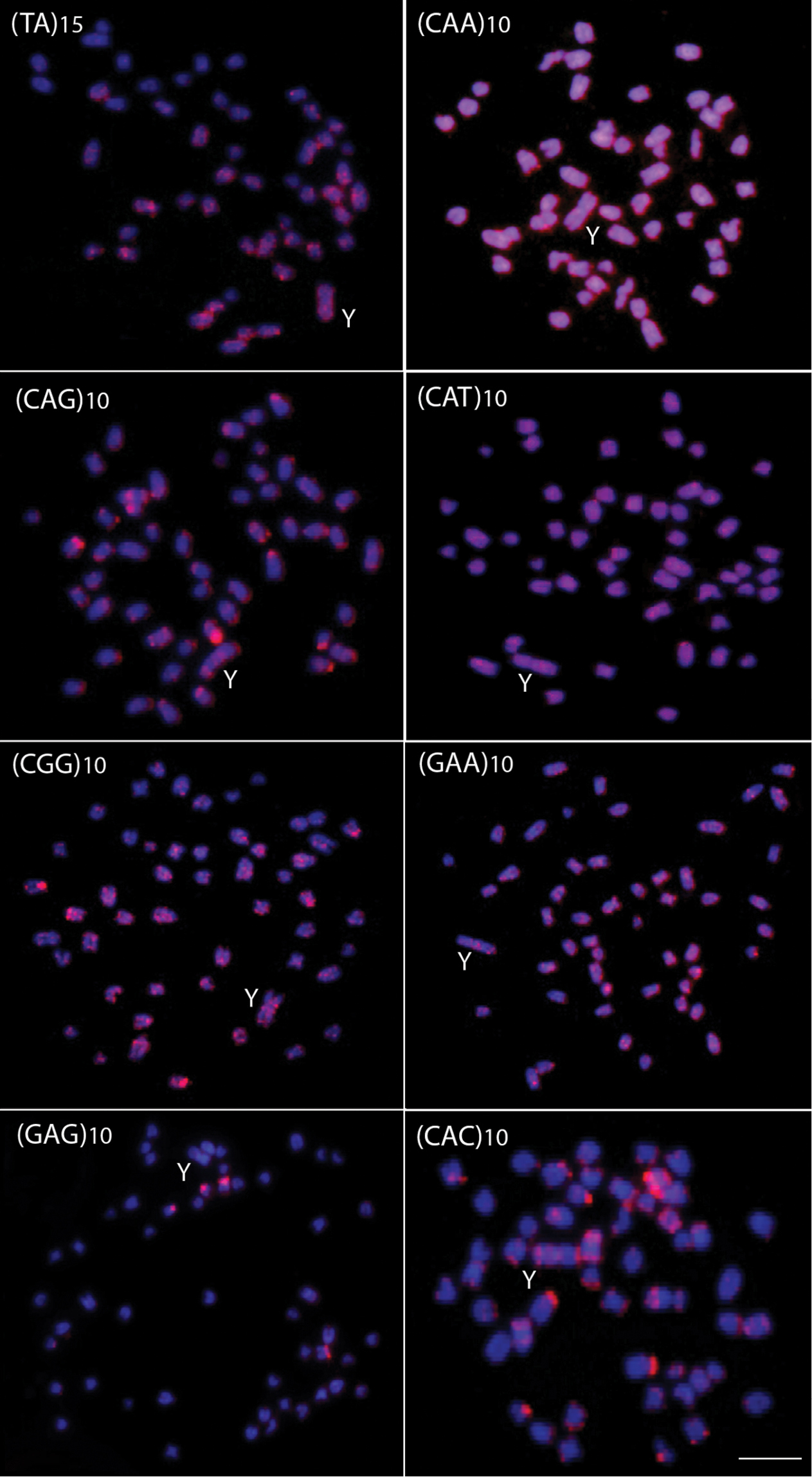
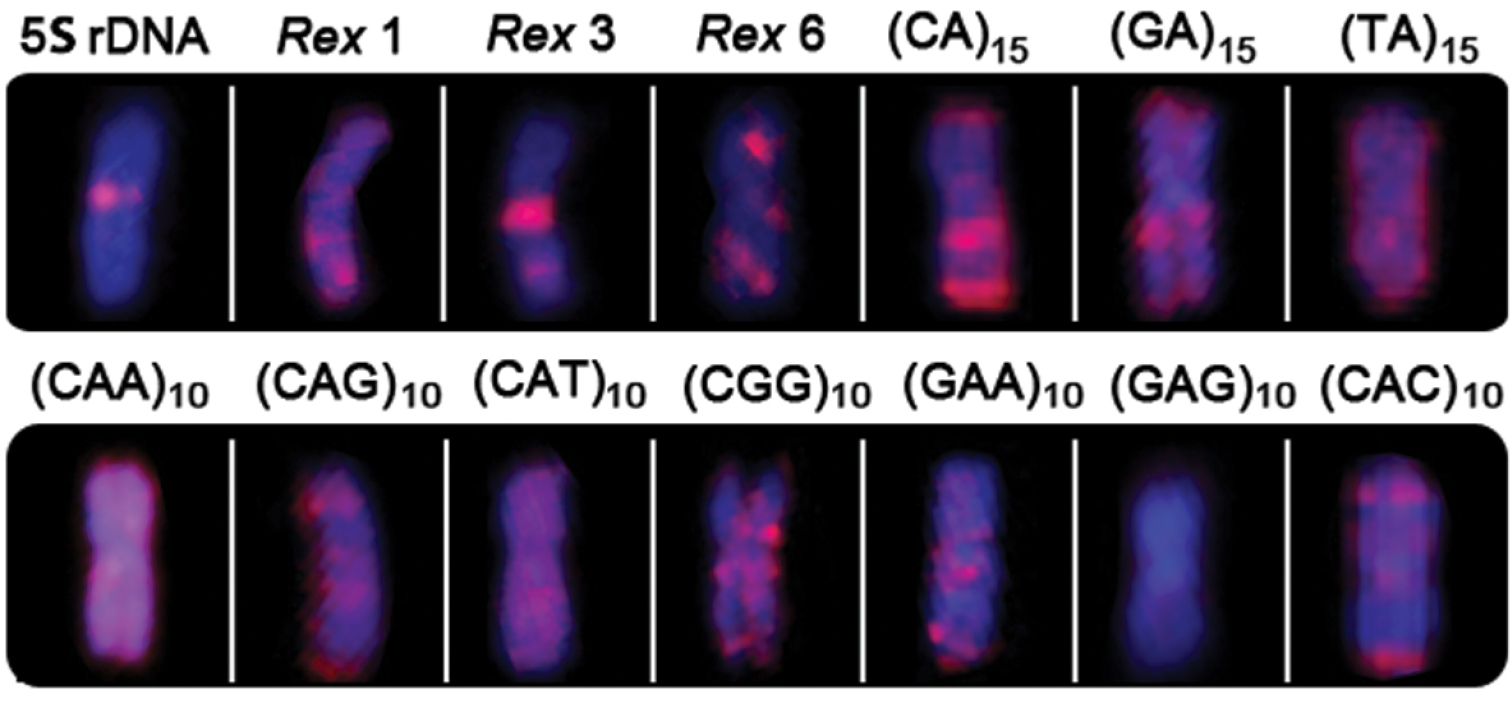
The microsatellite sequences show a differential distribution, some of them restricted to telomeric and/or interstitial regions and others with a scattered distribution on the chromosomes. Microsatellites (CA)15, (GA)15, (CAC)10 and (CAG)10 are mainly accumulated in the telomeric regions of the chromosomes and in some interstitial sites, but with a different distribution, since some chromosomes present higher signals than other ones (Figs 2 and 3). However, the (CA)15 sequences present a greater distribution compared with the other three classes of microsatellites, including on the Y-chromosome. In fact, this chromosome show a greater accumulation for the (CA)15 microsatellite, mainly in interstitial and telomeric regions of the long arms. In turn, the microsatellites (CAA)10, (CAT)10, (CGG)10, (GAA)10 and (TA)15 present a dispersed distribution among the autosomes and on the Y chromosomes (Fig. 3). In contrast, (GAG)10 microsatellite is poorly represented in the genome of Erythrinus erythrinus, with only four chromosomes showing mapped sites in their centromeric region. The Y-chromosome shows no labeling for this microsatellite (Fig. 3). Figure 4 highlights the distribution of all repetitive sequences analyzed along the Y-chromosome of the species.
Male metaphase plates of Erythrinus erythrinus probed with microsatellite sequences. Bar = 5 μm.
Distribution of repetitive DNA sequences in the Y chromosome of Erythrinus erythrinus.
The repetitive fraction of the genome can be a useful tool for the identification of recent genomic changes that occurred during the evolutionary process. Retrotransposons usually carry regulatory sequences and may attract methylation, thus influencing the gene expression (
The Rex family seems to be abundant in different teleost species (
In Erythrinus erythrinus (karyomorph C), Rex 3 showed a clear colocalization with 5S rDNA sites in the centromeric region of several chromosomes. Our data agree with previous results achieved for this same karyomorph (
Microsatellites mapping has shown both similar as well as different distribution patterns between species (
The cytogenetic mapping of repetitive DNAs has improved the knowledge of the evolutionary origin of the neo-Y chromosome. In fact, the chromosomal mapping of repetitive DNA sequences has shown differential accumulations on the sex-specific chromosomes (
In Erythrinus erythrinus, a centric fusion was proposed to be related with the origin of the big metacentric Y chromosome found in karyomorphs B, C and D and the differentiation of the X1X1X2X2/X1X2Y multiple sex system in these karyomorphs (
In turn, the mapping of microsatellites in the chromosomes has also been useful tools for analyzing the differentiation of sex chromosomes. In simple sex chromosomes, such as the ZZ/ZW system of Leporinus reinhardti Lütken, 1875 and Triportheus auritus (Valenciennes, 1850) (
Additionally, no preferential accumulation of microsatellites was found to occur in the sex chromosomes of Erythrinus erythrinus. In fact, there were no significant differences in the distribution of the microsatellites analyzed concerning to autosomes and sex chromosomes, based on the neo-Y chromosome which is easily identifiable in this species. It is well known that the suppression of the recombination is a crucial step in the differentiation of the sex pair, leading to the differentiation of the sex-specific chromosomes. Multiple sex chromosome systems originate from chromosomal rearrangements from simple systems and can itself reduce or eliminate the recombination near breakpoints, reinforcing previous suggestions that other events, such as accumulation of repetitive DNAs, may not be necessary for this process (
The repetitive sequences used in this study did not show a differential accumulation in the neo-Y chromosome of Erythrinus erythrinus, showing a similar distribution to the other chromosomes of the complement. However, it is clear that different repetitive DNAs may exhibit differential distribution patterns in chromosomes, including the neo-Y one (Figure 4), probably reflecting differences in the time of chromosomal occupation, as well as of strategies for dispersal throughout the genome.
This work was supported by the Brazilian agencies CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico), FAPESP (Fundação de Amparo à Pesquisa do Estado de São Paulo), and CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior).