Citation: Cristiano MP, Simões TG, Lopes DM, Pompolo SG (2014) Cytogenetics of Melitoma segmentaria (Fabricius, 1804) (Hymenoptera, Apidae) reveals differences in the characteristics of heterochromatin in bees. Comparative Cytogenetics 8(3): 223–231. doi: 10.3897/CompCytogen.v8i3.7510
To date, more than 65 species of Brazilian bees (of the superfamily Apoidea) have been cytogenetically studied, but only a few solitary species have been analyzed. One example is the genus Melitoma Lepeletier & Serville, 1828, for which there is no report in the literature with regard to cytogenetic studies. The objective of the present study is to analyze the chromosome number and morphology of the species Melitoma segmentaria (Fabricius, 1804), as well as to determine the pattern of heterochromatin distribution and identify the adenine–thymine (AT)- and guanine–cytosine (GC)-rich regions. Melitoma segmentaria presents chromosome numbers of 2n=30 (females) and n=15 (males). With C-banding, it is possible to classify the chromosomes into seven pseudo-acrocentric pairs (AM), seven pseudo-acrocentric pairs with interstitial heterochromatin (AMi), and one totally heterochromatic metacentric pair (Mh). Fluorochrome staining has revealed that heterochromatin present in the chromosomal arms is rich in GC base pairs (CMA3+) and the centromeric region is rich in AT base pairs (DAPI+). The composition found for Melitoma diverges from the pattern observed in other bees, in which the heterochromatin is usually rich in AT. In bees, few heterochromatic regions are rich in GC and these are usually associated with or localized close to the nucleolus organizer regions (NORs). Silver nitrate impregnation marks the heterochromatin present in the chromosome arms, which makes identification of the NOR in the chromosomes impossible. As this technique reveals proteins in the NOR, the observation that is made in the present study suggests that the proteins found in the heterochromatin are qualitatively similar to those in the NOR.
cytogenetic characterization, heterochromatin, fluorochromes, solitary bees, karyotype evolution
The genus Melitoma Lepeletier & Serville, 1828 belongs to the tribe Emphorini and has 10 described species. These are solitary bees, which nest in cavities in the soil, are typically gregarious, and are distributed from Mexico to Argentina (
Cytogenetic studies of Brazilian bees are more common in the eusocial species belonging to the tribe Meliponini. These studies were initiated by
Little cytogenetic information has been obtained for solitary bees. In the literature, only some cytogenetic information for the species of the genus Eufriesea Cockerell, 1908 (
Cytogenetic studies are important because they contribute a great deal to the understanding of evolutionary mechanisms that contribute to the changes in genome organization. By using different chromosomal banding techniques we can study different chromosomal characters that can be used to solve taxonomic issues. A simple karyotype analysis allows the observation of variations, such as, differences in chromosome number, size, and specific base pair composition of the DNA, enhancing our knowledge of the evolution and phylogenetic relationships of different species (
The “minimum interaction hypothesis” proposed by
In this context, the aim of the present study is to analyze the karyotype, including the chromosome number and morphology, distribution pattern of the heterochromatin, and richness of composition of the AT and GC base pairs, of the solitary bee species Melitoma segmentaria, thereby contributing to an increase in the cytogenetic knowledge of this genus and providing interesting new insights about the genome organization in these bees.
To perform the cytogenetic study, 10 larvae of Melitoma segmentaria within the nest cells were collected in Viçosa – Minas Gerais, Brazil (20°44'58.03"S, 42°51'8.98"W). We sampled 10 individual nests. The cells were opened in the laboratory to verify the larval stage. The larvae that were not at the post-defecation stage were maintained in a biological oxygen demand (BOD) chamber (Marconi, model MA-415/S), at 25°C, until they reached the appropriate stage.
The metaphase chromosomes were obtained from the larval cerebral ganglia in the post-defecation stage (
The metaphases were analyzed with the aid of an Olympus BX 60 microscope coupled to an image capturing system, Q Color3 Olympus®. For analysis of the fluorochromes, WB filters (450 – 480 nm) were used for CMA3 and WU filters (330 – 385 nm) for DAPI. The karyotypes were assembled according to the classification established by
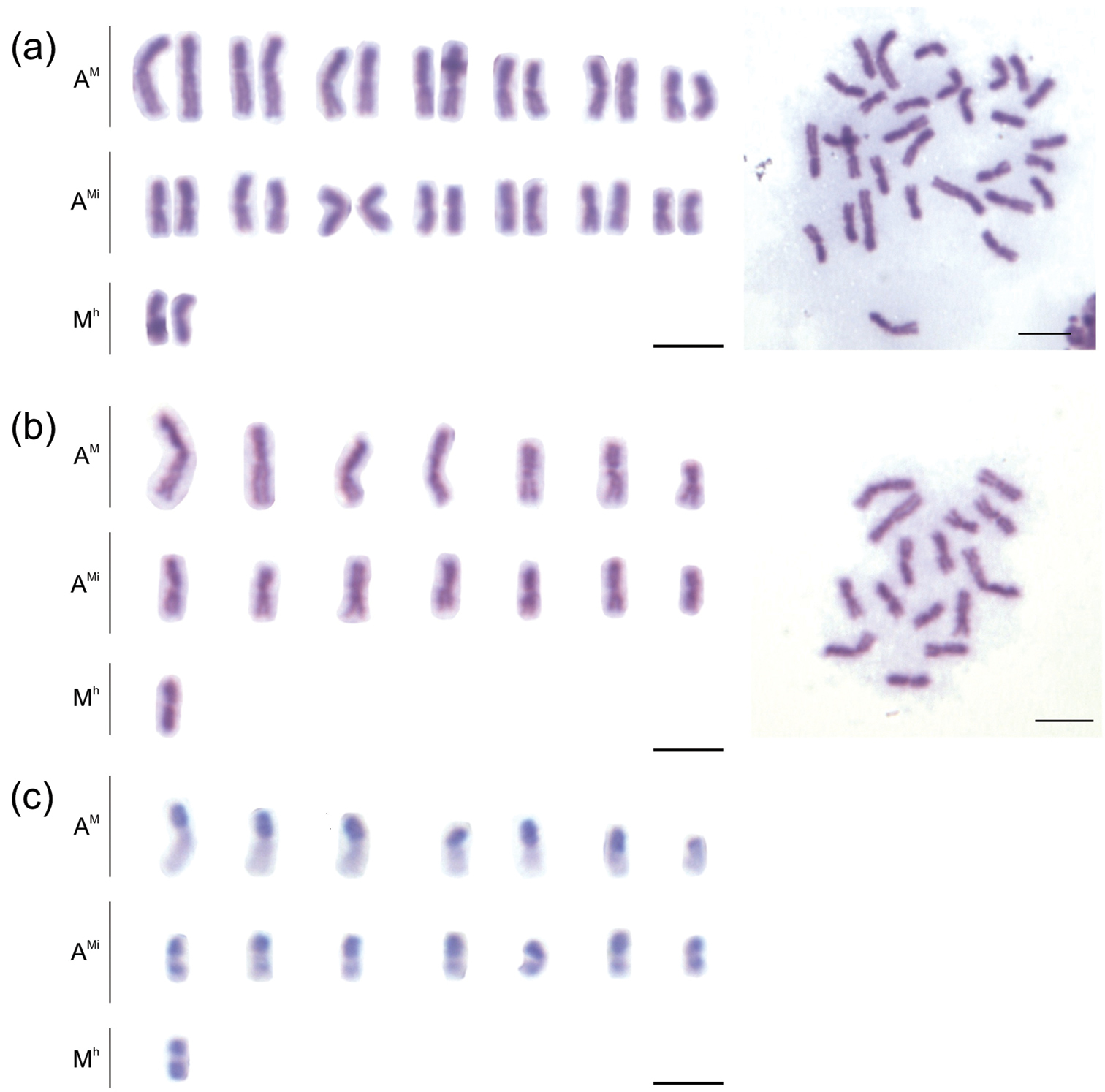
The species Melitoma segmentaria showed a chromosome number of 2n=30 for females and n=15 for males (Fig. 1). This chromosome number was similar to that observed in other solitary bee species, including Ceratina megastigmata Yasumatsu and Hirashima, 1969 (2n=34), Xylocopa appendiculata Smith, 1952 (2n=32), and Pithitis smaragdula (Fabricius, 1787) (2n=28) (
The C-banding technique allows the observation of large positive heterochromatic blocks in the chromosomes of Melitoma segmentaria (Fig. 1c), wherein, at least one of the arms, has been completely heterochromatic. Taking into account the C-banding pattern and the nomenclature proposed by
Mitotic karyotypes of Melitoma segmentaria. a Giemsa staining (female) b–c C-banding (male). Bar=5μm.
The pattern of heterochromatin distribution in Melitoma segmentaria is similar to that observed in most of the studied Meliponini species (
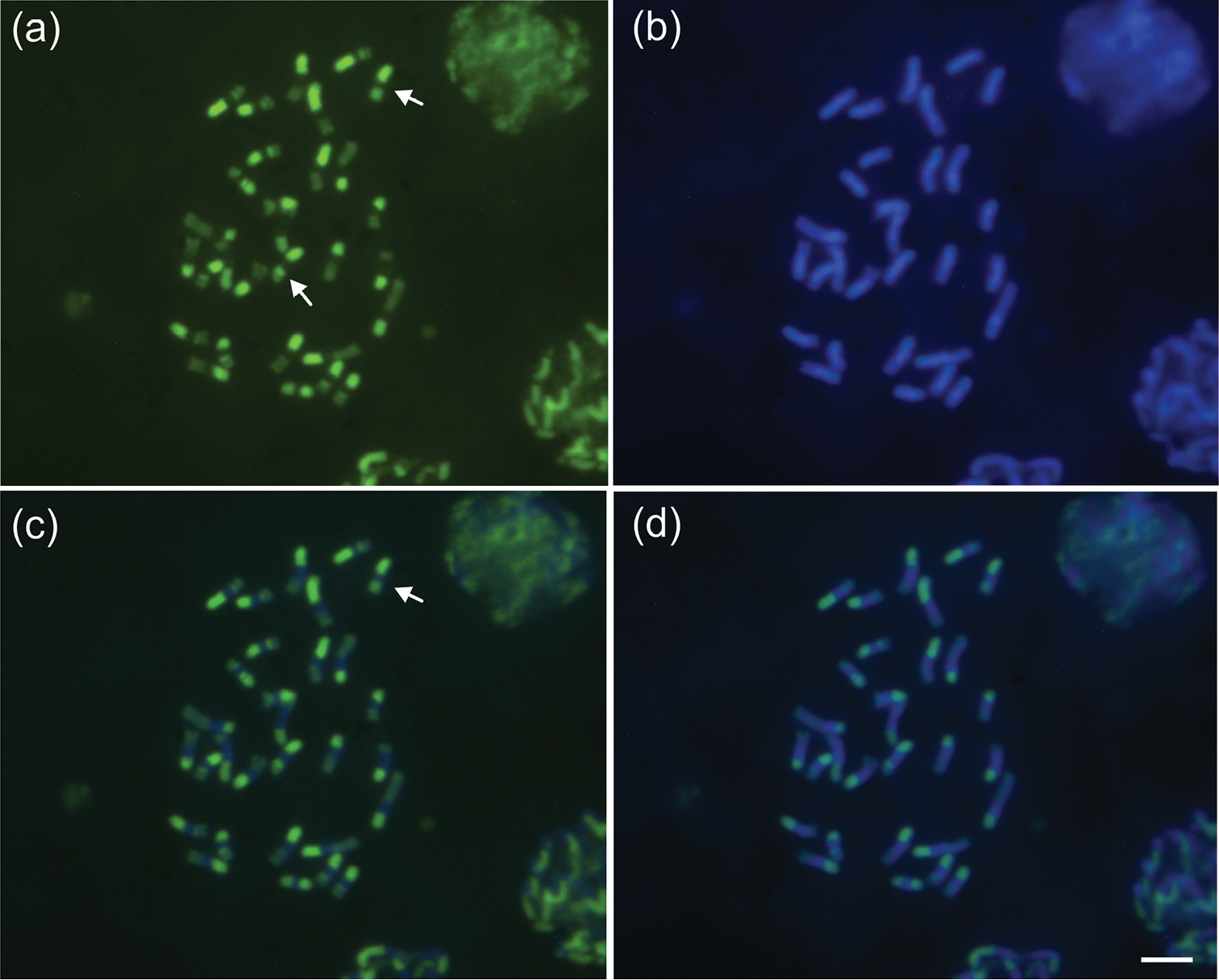
Chromosome staining with the fluorochromes CMA3 and DAPI (Fig. 2) shows that heterochromatin has an apparently homogeneous constitution. However, the fluorochrome CMA3 shows that the heterochromatin present in the chromosomal arms of Melitoma segmentaria is more GC-rich than AT-rich. DAPI in Melitoma segmentaria marked the centromeric and pericentromeric regions of the chromosomes, indicating that these regions are rich in AT base pairs. In Meliponini bees the heterochromatin is rich in AT base pairs (it is therefore DAPI+) (
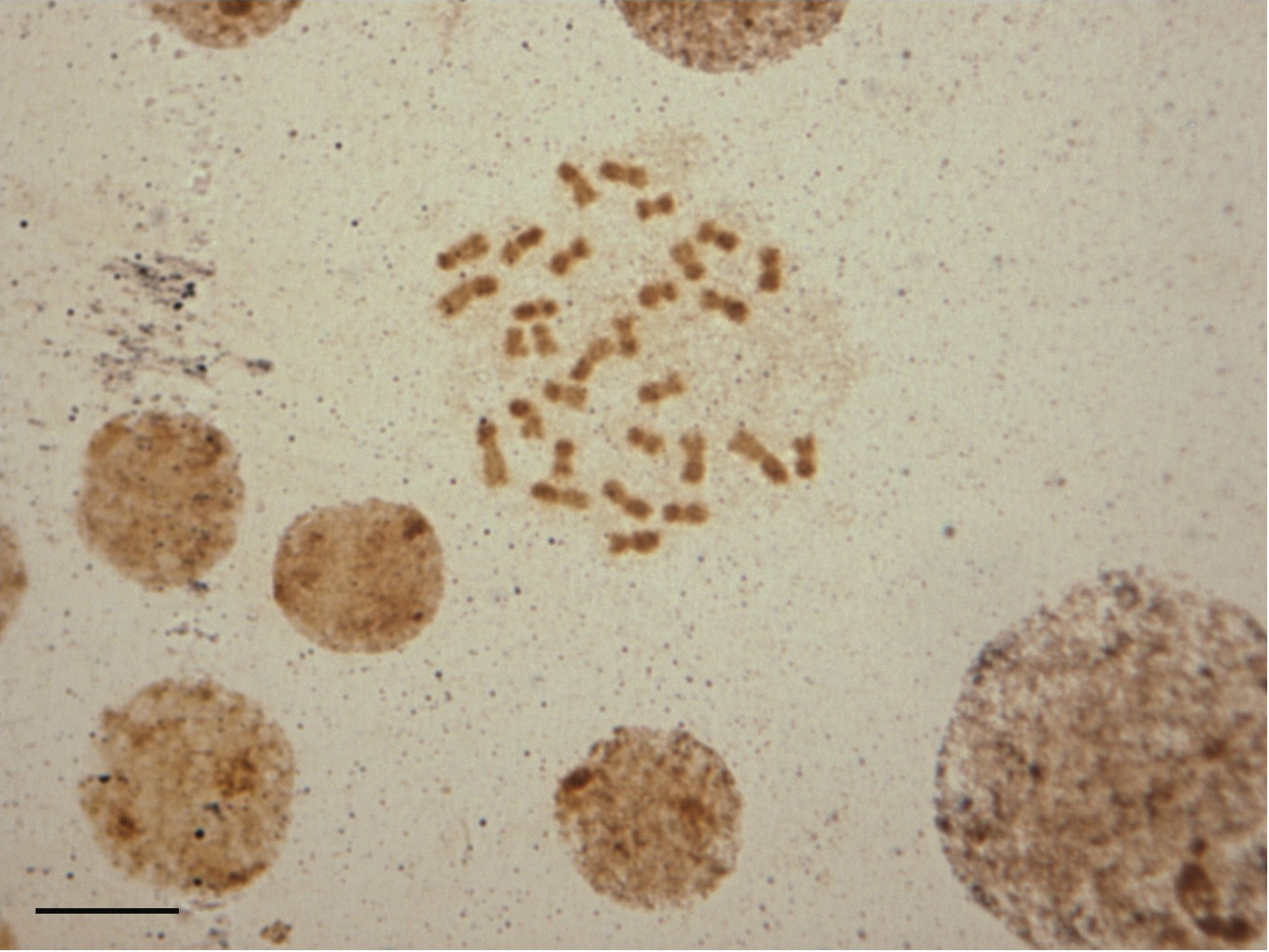
In order to identify the position of the NORs in the genome of Melitoma segmentaria, impregnation with silver nitrate was performed (Fig. 3). However, the methodology used was not efficient enough to indicate the location of the NOR. A particular pattern found in the chromosomes of Melitoma segmentaria was a result of silver staining of the heterochromatic chromosomal arms. Overall, NORs were associated with the GC-rich regions, as observed in the bee genus Friesella Moure, 1946 (
The silver impregnation technique located the NOR by staining the proteins present in this region.
This study is the first detailed karyotype characterization of the Melitoma species, bringing to light several chromosome features, such as, chromosome number, morphology, heterochromatin pattern, and base pair richness. Characterizations of the karyotype of other species of solitary bees and of the genus Melitoma, coupled with the use of banding and staining techniques are needed, to obtain a better understanding of the chromosomal evolution in Apidae.
Female mitotic karyotypes of Melitoma segmentaria stained with fluorochromes: a CMA3b DAPI c CMA3/DAPI and d DAPI/CMA3. Arrows indicate entirely heterochromatic metacentric chromosomes (Mh). Bar=5μm.
Female mitotic chromosomes of Melitoma segmentaria submitted to silver-nitrate staining. Dark regions on the heterochromatin arms indicate silver staining. Bar=5μm.
The authors would like to thank Prof. Lucio Antonio de Oliveira Campos for his suggestions and D.Sc. Danon Clemes Cardoso for the helpful comments on the manuscript. The authors also thank Fundação de Amparo à Pesquisa de Minas Gerais (FAPEMIG) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for the financial support.